Understanding AAMI Level 4 — The Benchmark for Barrier Integrity
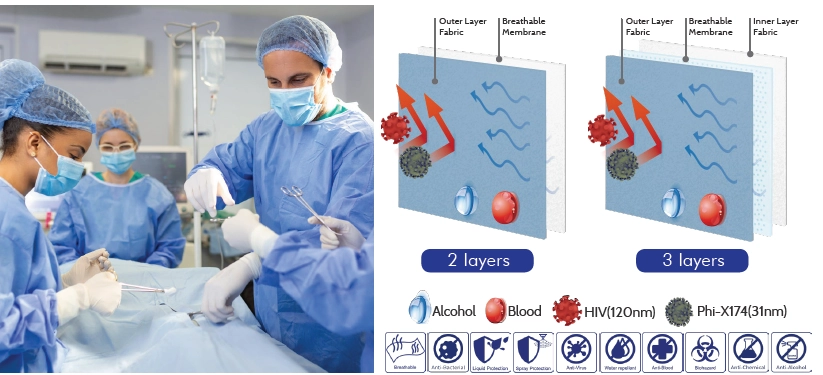
AAMI Level 4 is the highest protection level under the AAMI PB70 standard.It is assigned to materials used in high-fluid, high-infection-risk procedures, including orthopedic, cardiovascular, and trauma surgeries.
To meet Level 4 requirements, the fabric must pass ASTM F1671 viral penetration testing—the defining threshold separating Level 3 and Level 4.
Why Kae Hwa Is Trusted in Medical Barrier Materials
With over 60 years of polymer and lamination expertise, Kae Hwa is a proven specialist in medical barrier fabrics used in high-risk surgical and protective applications. Our materials are internationally certified—including AAMI Level 4, ASTM F1671/F1670, EN 14126, and CE Type 4B/5B/6B—ensuring reliable viral, fluid, and pathogen protection for clinical environments.
We develop medical laminates using proprietary technologies such as MicroBreath™ microporous membranes, AquaVene™ monolithic hydrophilic films, and HydraProof™ dense waterproof barriers, enabling both strong protection and sustained comfort.
About Us